October 28, 2021
Focus on the future of validation and customized validation services drive further efficiencies
MORGAN HILL, CA – October 26, 2021 – AssurX, Inc. (AssurX), a leader in quality and compliance management solutions for regulated industries, announced today that it has further enhanced its QMS validation solutions portfolio. The improvements support regulatory guidance for AssurX QMS software validation while increasing efficiency and cost-effectiveness.
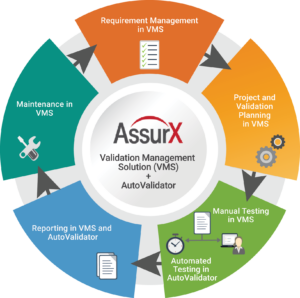
The AssurX QMS Validation Management Solution (VMS)
The VMS modernizes software development and validation lifecycles with a paperless process for managing requirements, performing risk assessments, testing, and reporting. The result is a faster speed to completion which improves quality and reduces validation costs.
The AssurX VMS creates a consistent and standardized computer system validation process that prevents data integrity issues. In addition, VMS eliminates the need for time consuming activities including paper-based validation, printing, signing and manual tracking.
“Using a paper-based validation process is error-prone and time-consuming. Plus, there are high operational costs with handling paper-based records, traceability issues, no reusability of artifacts, and limited visibility into validations of similar systems,” said Victoria Alestra, Software Quality Assurance Manager at AssurX. “AssurX VMS considerably reduces effort and increases the audit-readiness by gaining control over the development and validation process by automating deliverables.”
AssurX Validation Management Solution deliverables include:
- User and Functional Requirement Specification
- Risk Assessments
- Requirement Traceability Matrix
- Validation Plans
- Validation Summary and Detailed Reports
- Test Protocols
The AssurX AutoValidator
In addition to VMS, AssurX released the in 2019, a tool that automates the execution of validation tasks to ensure software systems are in a constant state of control. The AssurX AutoValidator allows you to take the OQ/PQ scripts and run them in the exact same way a manual script would be run, but much faster. While the majority of manual executions typically take a full week to complete, the AutoValidator can run the same scripts in only a few hours. The AssurX AutoValidator leaves no stone unturned. Every object, action and expected result that is recorded is accounted for, which eliminates common human errors such as missing a step or inputting inaccurate data during validation. A comprehensive report is generated displaying the pass and fail areas including screenshots before execution and after.
The powerful combination of the AssurX Validation Management Solution and the AutoValidator produces a streamlined, paperless method that can adapt to any validation framework. Customers can conduct both manual and automated testing, complete requirement traceability, increase visibility on configuration issues, and generate detailed validation reports.
Risk-Based Validation
Validation is fully aligned with the FDA’s shift to risk-based Computer System Assurance (CSA). CSA encourages the use of automation for software testing and leveraging vendor documentation and validation activities. AssurX has developed a double-V model for QMS validation that provides customized validation services based on a critical thinking, “least burdensome approach” that supports the GAMP 5 Model.
AssurX Risk-Based Validation Packages reduce the overall computer system validation project time by leveraging AssurX’s extensive experience and complete packages, helping to decrease production time without compromising compliance or quality.
Combine Validation Solutions to Maximize Audit Readiness
The risk-based validation packages seamlessly integrate with the Validation Management Solution and AutoValidator to create an end-to-end software and validation processes for audit readiness and compliance.
“Leveraging AssurX Risk-Based Validation Packages and AssurX Validation Services has decreased our customers’ go-live time as much as 70%. Applications like FDA eMDR and EU MDR/IVDR, or applications where minimal configuration changes typically occur, enable our customers to be audit-ready after the initial installation of the application,” said Alestra. “Validation is necessary and doing it right from the beginning is the best way to ensure a continual state of validation.”
Benefits of AssurX Validation Solutions
While computer systems validation is often seen as costly and compulsory, there are significant benefits beyond FDA compliance. AssurX reduces the burden of validation with notable organizational benefits that include:
- Gain greater insight: Allow every individual, team, and organization to have full visibility into projects, reports, and items across the application lifecycle.
- Improve collaboration: Bring together teams to participate in planning, requirement gathering, making changes, viewing tasks status and other vital project information in one central location.
- Increase productivity: Using the solution implements and enforces best practice processes and proven methodologies that drive improvement in productivity.
- Improve Quality: Reduce the number of defects due to miscommunication, identify inconsistencies between requirements, enable efficient testing, and ensure that the final application meets the needs and expectations of the user.
Validation Services for any Environment
“AssurX enables any organization, of any size, in any location, to conduct validation based on their quality environment and team capabilities,” explained Alestra. “Not all customers have in-house expertise or a dedicated staff for validation. In that case, the AssurX Validation Services team can operate independently from the implementation services team. This approach eliminates any perceived bias in the validation process and the cost and effort of hiring and training testers.”
Future Enhancements
AssurX Validation Management Solution and Services is actively monitoring the FDA’s Computer Software Assurance guidance closely and continuing to further align with CGMP. AssurX’s progressive solutions and services will lead to increased awareness and knowledge about CSA principles and benefits and can help teach and mentor your team on CSA principles and how to apply critical thinking to your process. Furthermore, Validation Services has the ability to assess your CSV process and recommend changes based on your quality of documentation, testing, SOPs/WIs, testing, use of automation, and performance on audits.
RELATED READING: Computer Systems Assurance: Making the Transition from Computer Systems Validation