Deviation Management Software for Regulated Companies
AssurX Deviation Management Software provides an automated solution to capture, investigate, and disposition planned and unplanned deviations and nonconformances. Fully configurable, best practices workflows speed the identification of the deviation source anywhere in the quality value chain. Track and manage a wide range of deviations from initiation, through investigation and root cause analysis, to CAPAs and resulting change controls.
Understand and control process deviations and product nonconformances to avoid regulatory action and financial losses that can compromise to your brand. Maintain compliance with regulations and standards for deviations and nonconformances including, but not limited to:
AssurX centralizes deviation management by providing a system of record for capturing, investigating, and resolving planned and unplanned deviations, while delivering insight into trends that support continuous improvement and product quality.
Comprehensive Deviation Management Features and Functionality
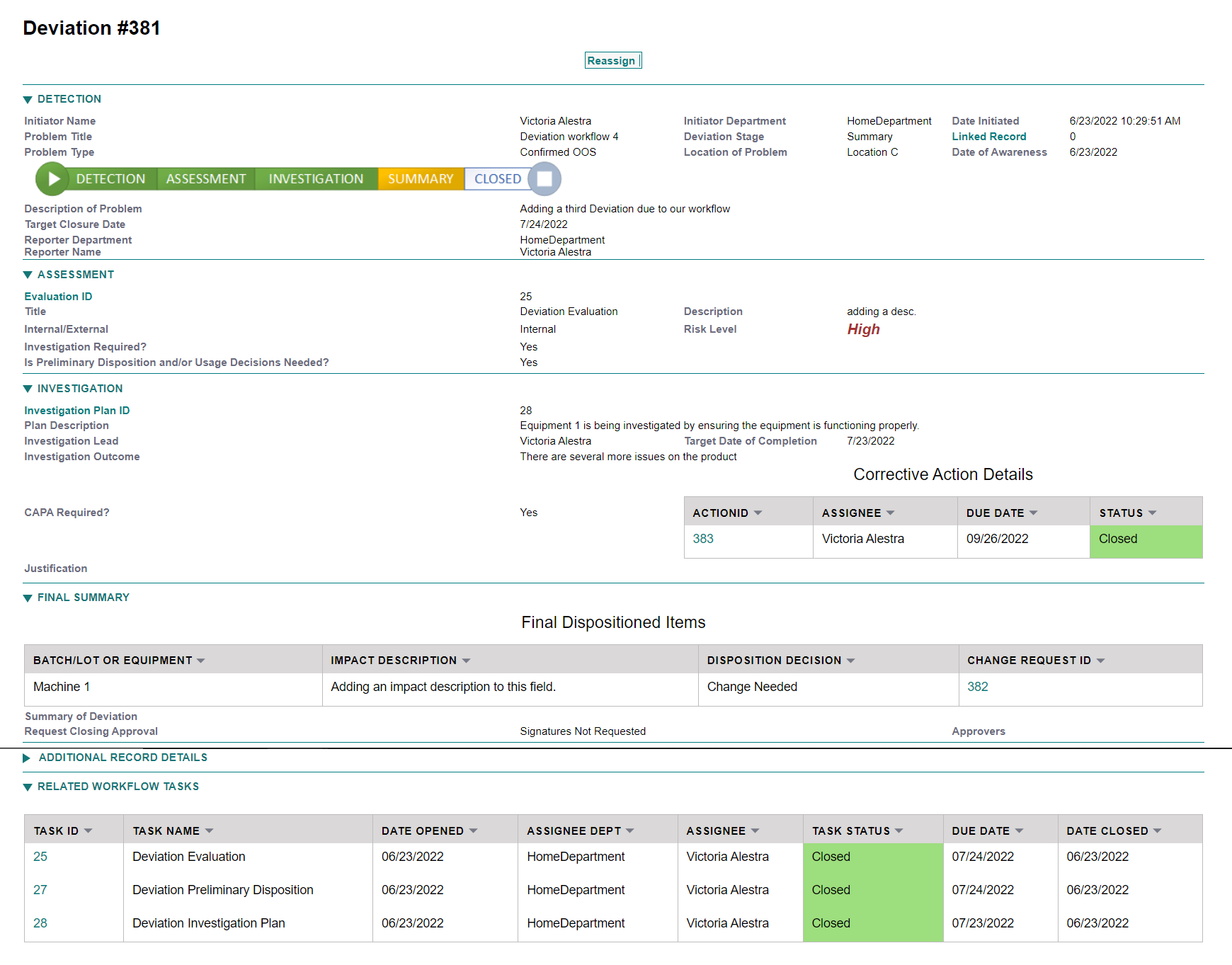
AssurX enables rigorous and timely management of deviations with configurable workflows that align with your business needs. As a result, deviation investigations follow a pre-defined process that improves time to resolution and compliance posture, as well as gain insights that enable you to continually improve processes and product quality. Streamline the deviation management process, minimize errors, and facilitate compliance.
Integrate deviation management software with external enterprise applications including ERP, MES, batch release systems, and other systems with a robust, open interface. Exchange information bi-directionally with suppliers, development, and other departments.
PLATFORM
AssurX core quality processes are available to you from day one. This allows your organization to build out and integrate processes as your quality plan expands or as change requires.
CONFIGURABILITY
Highly configurable, zero-code process workflows provide an environment that is scalable and flexible to meet changing business needs.
EFFICIENCY
Automated alerts, escalations, dashboard functionality, and extensive reporting capabilities keep the process moving on-time with enterprise-wide visibility.
ACCOUNTABILITY
Integrated tasks, assignments, signatures, and audit trails keep your users accountable and in compliance with regulatory guidelines.
STANDARDIZATION
Decrease the likelihood of repeat errors with the help of built-in decision trees and drill-down selections for multi-level investigations and root cause analysis.
FMEA–ROOT CAUSE ANALYSIS
A process-driven, step-by-step failure mode and effects analysis (FMEA) is available within AssurX to identify all potential causes of a deviation.
RISK CONTROLS
Data entered into the deviation record generates a risk priority number (RPN) based on your designated risk factors that include severity, occurrence, and other factors.
VISIBILITY
Configurable dashboards, reports and analytical data provide clear insights in the status of open deviations, trends, and the effectiveness of risk management.
Watch a 6 Minute Video of the AssurX Deviation Management Solution Now
See how the AssurX Deviation Management Solution provides a single source of truth to capture, investigate, and correct deviations, regardless of source or type. Workflows are also highly configurable to adapt to the specific needs of your organization.
Benefits of AssurX Deviation Management Software
TRUSTED BY GLOBAL COMPANIES FOR OVER 20 YEARS
AssurX: Connected Quality and Validated Compliance
AssurX provides expert technical and program management oversight for customer installs around the globe. We use a proven implementation process that provides a consistent method for architecture design.
DATA MIGRATION
Our experts are experienced with a host of different methods for performing migrations, from direct migration using AssurX import tools, to building data warehouses that used for robust reporting.
AssurX: Quality & Compliance Systems for Every Enterprise
The quality and compliance journey can be an obstacle course of changing regulations and standards in a dynamic global market. The AssurX enterprise quality management system (EQMS) and the AssurX Energy Compliance System (ECOS) help guide organizations through the complexities of any regulatory landscape.
Our dynamic, connected quality and compliance management solutions help guide your journey by automating processes to streamline event management, minimize risk, measure performance and facilitate change. The result is a proactive system that improves efficiency, compliance readiness, product quality, and customer satisfaction.






One Platform. Every Solution.
AssurX centralizes quality management and regulatory compliance processes within a single, configurable software platform. The system includes core quality and compliance workflows out of the box, eliminating the need for separate modules and reducing reliance on paper-based or disconnected systems.
Organizations can deploy the solutions they need today and add additional capabilities as requirements evolve. With pre-configured workflows, searchable data, and flexible integration options, AssurX supports efficient deployment, configuration, and long-term scalability across the enterprise.