Manage, review, and document controlled changes across quality and compliance processes.
As they say, change is the only thing constant, even in regulated environments where uncontrolled changes can jeopardize product safety and reliability. For regulated companies, any change in equipment, product materials, design specifications, formulations, processes, and quality documentation must be reviewed, investigated or tested, and approved. For this reason, change control is a critical element of quality management.
AssurX Change Control Management simplifies change control by automating and streamlining every step of the process, from change request and review through planning, approval, and implementation.
How AssurX Change Control Management Can Help You
The concept of a change control system in pharmaceuticals, medical device, diagnostics, and other FDA-regulated sectors is an inherent part of regulatory compliance. AssurX is designed to address change control requirements found in 21 CFR Part 820, Current Good Manufacturing Practice (CGMP) and 21 CFR Parts 210-211, ISO 9000 series, and ISO 13485 standards.
AssurX makes change control systematic and compliant. The following features facilitate a faster and compliant process:
Transform Change Control Management with AssurX
Many organizations cling to archaic, unconnected electronic systems or even paper-based processes due to budget concerns. They endure inefficiency, lack of transparency, and the risk of noncompliance. Modernize your change management process through an automated, interconnected system. With AssurX, you’ll enjoy the following benefits:
Increased Accountability and Transparency
Change tasks are automatically routed and escalated so they can’t fall through the cracks. Audit trails make it easy to see and track down who did what and when.
Effective and Convenient Collaboration
AssurX provides robust tools to facilitate collaboration, from change submission and review to planning of proposed changes to tracking of feedback and all the way through change approval and implementation.
A Single Source of Truth
Integrate your quality processes with change management quality-related tasks and activities. Change control management will be seamlessly connected with CAPA, audit, deviation, supplier management, and other quality processes.
Cost- Effective QMS Change Management System
AssurX QMS change management software is available as a cloud-based or on-premises solution, reducing infrastructure and maintenance costs through centralized, automated workflows. Organizations can deploy on-premises today and migrate to the cloud later without disruption, as both deployment options share the same platform.
SIMPLE AND INTUITIVE INTERFACE
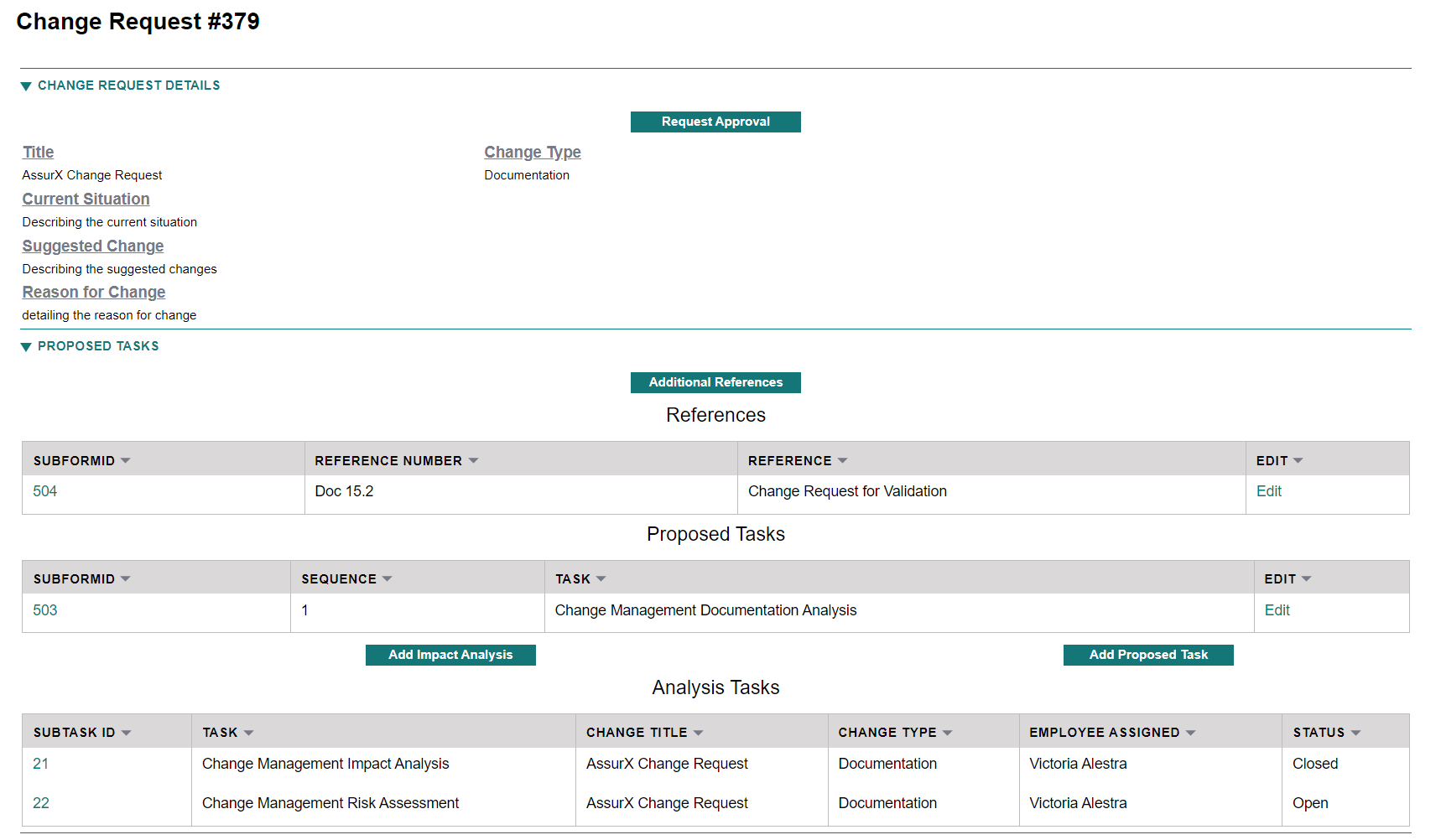
We’re not here to complicate things. We’re here to make your job much easier. AssurX’s Change Management Software’s change request form is simple, clean and easy to use and won’t require a colossal amount of time to train your users.
AssurX: Quality & Compliance Systems for Every Enterprise
The quality and compliance journey can be an obstacle course of changing regulations and standards in a dynamic global market. The AssurX enterprise quality management system (EQMS) and the AssurX Energy Compliance System (ECOS) help guide organizations through the complexities of any regulatory landscape.
Our dynamic, connected quality and compliance management solutions help guide your journey by automating processes to streamline event management, minimize risk, measure performance and facilitate change. The result is a proactive system that improves efficiency, compliance readiness, product quality, and customer satisfaction.






One Platform. Every Solution.
AssurX centralizes quality management and regulatory compliance processes within a single, configurable software platform. The system includes core quality and compliance workflows out of the box, eliminating the need for separate modules and reducing reliance on paper-based or disconnected systems.
Organizations can deploy the solutions they need today and add additional capabilities as requirements evolve. With pre-configured workflows, searchable data, and flexible integration options, AssurX supports efficient deployment, configuration, and long-term scalability across the enterprise.





















