The AssurX Medical Device Quality Management System: Aligning Quality Objectives with Compliance Requirements
The medical device market is full of opportunities to create life-saving products that diagnose and treat patients worldwide. AssurX medical device quality management system is designed to support manufacturers in marketing safe and effective medical devices. Additionally, it demonstrates compliance to meet those demands.
Device and diagnostics manufacturers use the AssurX EQMS to bridge the gap between aligning quality processes to improve product safety. They also maintain medical device compliance with applicable regulations.
TRUSTED BY
As an FDA-regulated company, Keystone Dental recognized the need to move beyond paper-based complaint handling processes that were hurting efficiency and increasing the complexity of reporting. By automating complaint management with AssurX, Keystone Dental was able to increase efficiency and accuracy while reducing the backlog of complaints and time to closure.
More Medical Device Success Stories
AssurX medical device quality management software helps enforce compliance with the following regulations and standards:
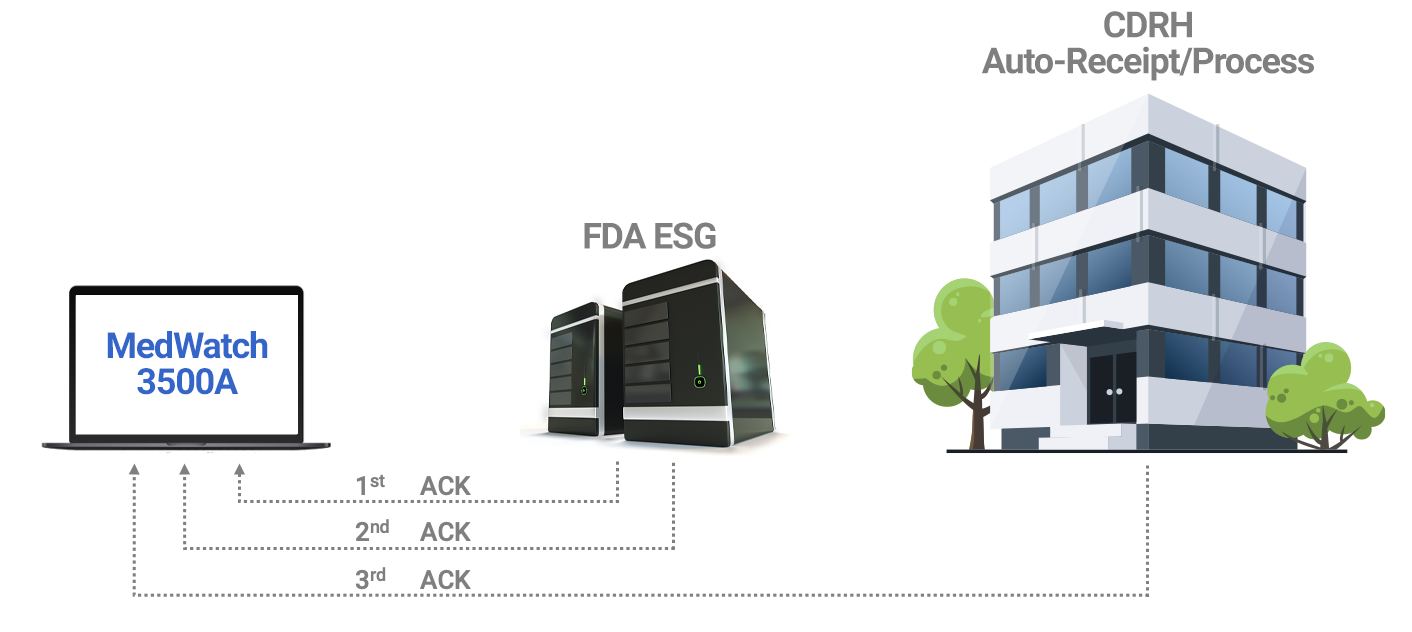
AssurX Medical Device Quality Management System Features
Manufacturers in the medical device industry and the diagnostics industry must maintain a laser focus on continuous quality improvement. Managing and controlling large volumes of documents, such as SOPs , training records, and quality manuals, is critical. A modern quality system using paper-based processes and siloed data is no longer viable.
Medical device QMS software facilitates compliance with stringent regulations by providing a framework for standardizing processes and maintaining proper documentation. Medical device compliance software automates document control, so teams use current and approved documents. This allows easier updates and audits for transparency and consistency.
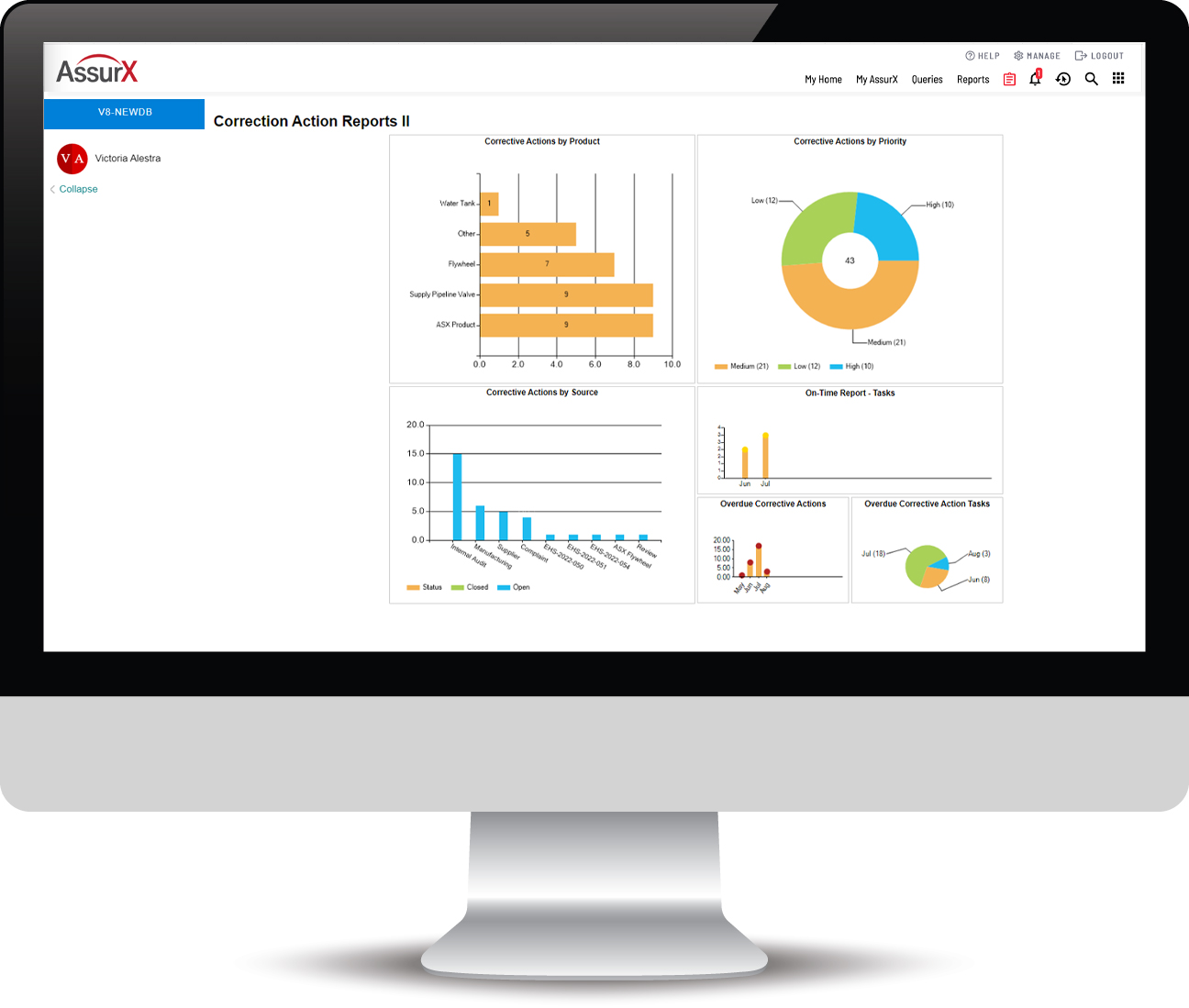
AssurX is built with robust features based on over 20 years of best practices, including:
Overcome Today’s Biggest Challenges for the Medical Device Industry
AssurX medical device quality management system accelerates your quality oversight to correct issues faster and implement preventive measures to prevent recurrence. Maintain a risk-based, proactive posture for efficient problem resolution.
Increased Efficiency
AssurX increases efficiency by integrating processes to close the loop on quality issues faster and more confidently. Launch CAPAs at any point in the quality chain. Our medical device QMS software can integrate risk management into all quality processes, ensuring a consistent approach to risk across the organization.
Quality Aligned with Compliance
AssurX makes it easier for your teams to participate in compliance efforts by incorporating regulatory requirements in your system. Configure the system to your organization’s unique medical device regulatory compliance demands. Our medical device compliance software helps you to be audit-ready. Its robust features provide comprehensive tracking and documentation capabilities.
Controls for HIPAA
Electronic PHI is accessible only to those who are allowed to see it. AssurX QMS software gives you the capability to set strict permission rights and shield unauthorized users from records that hold PHI.


Greater Control of Nonconformance Process
AssurX medical device manufacturing software provides a dynamic process for identifying, evaluating, documenting and disposition of non-conforming products as required per FDA 21 CFR 820 and ISO13485. Common terminology, failure codes, and escalation rules enable medical device and diagnostics manufacturers to identify and investigate the root cause of the issue diligently. They also demonstrate control of the non-conforming product, and use non-conformance data to help improve product quality at the design stage.
AssurX Complaint Management Software is a robust system designed to document, review, and report applicable adverse events to the FDA and other international bodies. Metrics & KPIs allow you to make informed decisions based on real-time data.
AssurX: Connected Quality and Validated Compliance
AssurX provides expert technical and program management oversight for customer deployments around the globe. We employ a structured implementation process, providing a consistent method for architecture design within our medical device quality management system.
AssurX: Quality & Compliance Systems for Every Enterprise
AssurX has been serving the medical device and diagnostics industry for more than two decades. Companies worldwide turn to us for help in reducing regulatory burden by automating quality processes while also demonstrating medical device regulatory compliance.
Our experts built an eQMS that can scale with your organization as it grows. Businesses need to shift and adapt frequently in the medical industry to stay competitive and our system allows that.
We can help you establish a new medical device quality management system or improve your existing system. Using unconnected ERP, PLM, and other enterprise applications? We can integrate them for a holistic approach to medical device regulatory compliance and optimal usage.






One Platform. Every Solution.
AssurX centralizes quality management and regulatory compliance processes within a single, configurable software platform. The system includes core quality and compliance workflows out of the box, eliminating the need for separate modules and reducing reliance on paper-based or disconnected systems.
Organizations can deploy the solutions they need today and add additional capabilities as requirements evolve. With pre-configured workflows, searchable data, and flexible integration options, AssurX supports efficient deployment, configuration, and long-term scalability across the enterprise.
































