Closed-Loop CAPA Management Software
Using AssurX, you can automate all the steps required to evaluate and investigate deviations. You can also implement the appropriate actions and controls to prevent further quality incidents. Corrective and preventive action software can help you streamline your quality management process.
AssurX CAPA management software also helps you track improvements and perform thorough root-cause analyses. This is one of the key features of our corrective action tracking software. In addition, the tightly integrated corrective and preventive action (CAPA) solution integrates with additional processes, data, and documents to drive quality improvements even further.
AssurX CAPA management software enables your enterprise quality management system to be self-correcting. It guides your organization from incident capture to closure while creating continual improvements in your quality chain. In addition, effectiveness checks and compliance verification with corrective action management software gives you confidence in your ISO and GxP alignment.
For organizations subject to FDA requirements, AssurX CAPA software supports the management of corrective and preventive actions in alignment with FDA regulations.
AssurX corrective action software system is designed to address regulatory requirements in regulations such as:
CAPA Software for Continuous Quality Improvement
CAPA management software is a modern essential for regulated companies. More importantly, it’s a basic element of continuous quality improvement. AssurX connects the CAPA process with document control, change management, training, deviation, non-conformance, audit, and other quality processes.
AssurX CAPA software provides a centralized platform and robust tools that can enable your organization to:
- Capture and manage CAPA records originating from audits, deviations, complaints, nonconformances, incidents, and risk assessments
- Conduct structured root cause analysis using configurable investigation workflows and recognized methodologies (e.g., fishbone, 5 Whys)
- Define, assign, and track corrective and preventive actions with clear ownership, due dates, escalation, and approval controls
- Assess and document risk and impact associated with identified issues, actions, and proposed changes
- Verify and document CAPA effectiveness through defined effectiveness checks and follow-up activities
- Maintain complete traceability between CAPAs, related deviations, complaints, risks, changes, and supporting documentation
- Support regulatory compliance and audit readiness with controlled records, approvals, and full audit trails
- Analyze trends and recurring issues to identify systemic problems and drive continuous improvement
- Reduce the likelihood of product recalls by identifying and addressing root causes before issues escalate
- Reduce the frequency and severity of customer complaints through consistent investigation and corrective action
- Reduce scrap and rework by addressing process, design, and supplier-related root causes
- Reduce product returns and minimize warranty-related costs by improving issue resolution and prevention
- Facilitate design and engineering changes by linking CAPAs to change management, design control, and validation processes
- Standardize CAPA processes across the organization while allowing configuration to meet specific regulatory, operational, or industry requirements
How AssurX Modernizes Your CAPA Management Process
AssurX CAPA software replaces paper-based and disconnected electronic systems with a centralized, automated platform for managing corrective and preventive actions. By integrating CAPA with related quality processes, the solution helps reduce manual effort and operational costs while supporting consistent, compliant execution through the following features and benefits:
Compliance Excellence
An end-to-end approach to CAPA actions provides significant compliance advantages. If process gaps are found at any time during the CAPA process, a change record can be launched. If documentation needs to be updated, AssurX Document Management gets the revision completed and approved. It can also can trigger a training task.
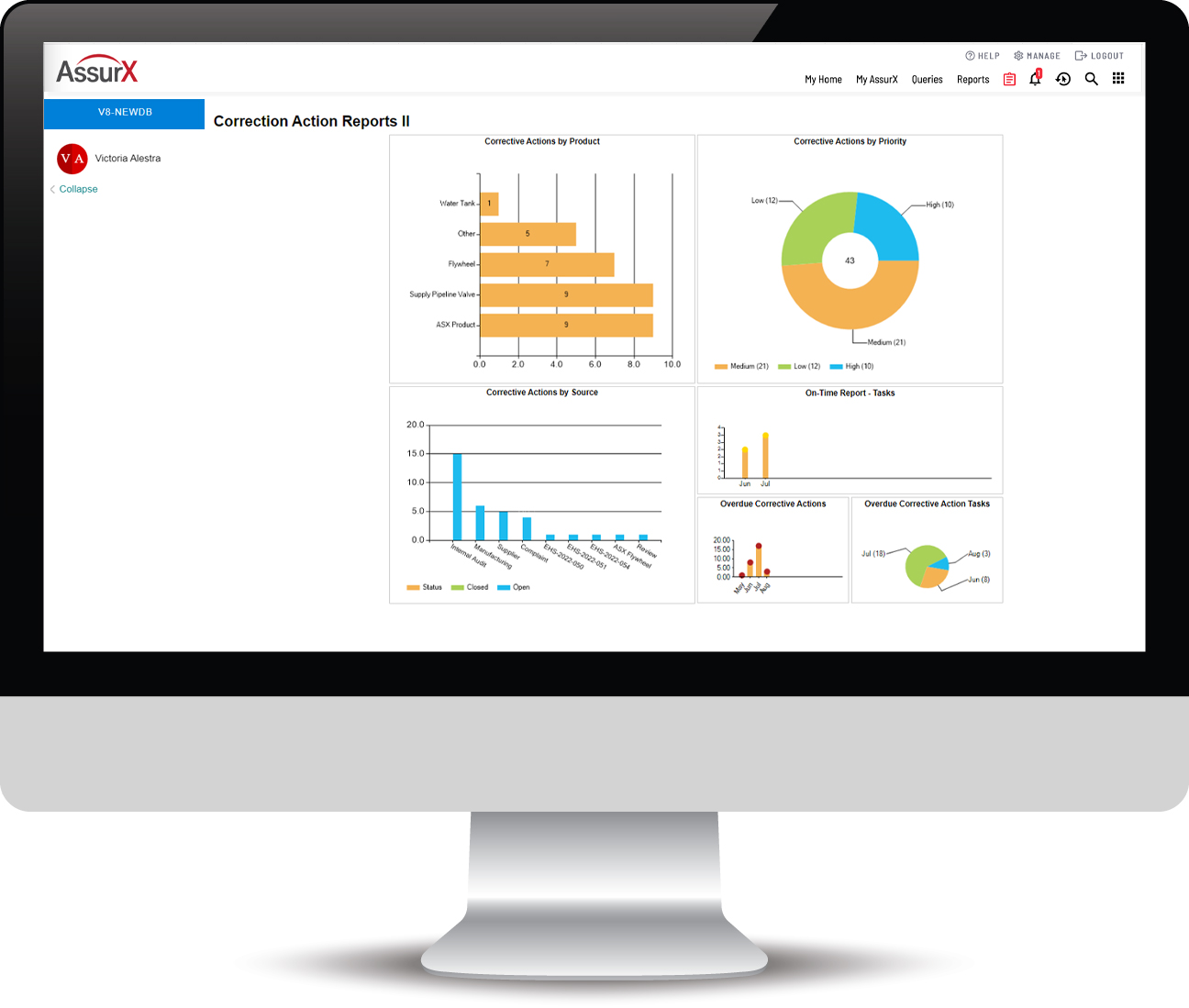
On-Demand Data
Provides efficient data and document retrieval and management. Reporting includes the ability to display specific records, including any attached images. Always know exactly where you are in the CAPA process. Trending and data analytics provide intelligence to pinpoint related issues and take proactive measures to address them.
Integration
CAPA software can be launched anywhere in your electronic quality chain. As a result, audits, non-conformances, deviations, supplier quality issues, and customer complaints all connect in a closed loop. With AssurX, you’ll be able to clearly determine that you have a quality issue. Our CAPA management software can then seamlessly trigger a CAPA.
Data Maximization
AssurX integrates with other business applications to provide greater insight and collaboration (for example, in CAPA investigations). Connect with MES, LIMS, ERP, CRM, and others to integrate your applications under a single CAPA software. This allows you to engage with other business areas seamlessly.
Centralization
Automate all CAPA-related tasks (routing, follow-up, escalation), under a centralized CAPA system to eliminate process vacuums.
Efficiency
AssurX CAPA management software speeds your time to CAPA completion. Tracking, automated reminders, escalations, and configurable dashboards keep assigned users on-path to task closure.
Auditability
AssurX CAPA software captures all related CAPA details including signatures and documentation. It creates a controlled way to organize report findings and attach related evidence records in one place.
Control Risk
A risk-based approach facilitates identification, analysis, and prioritization of risks in our CAPA software. As a result, the appropriate actions can be taken to proactively control and minimize the chances of future negative outcomes.






One Platform. Every Solution.
AssurX centralizes quality management and regulatory compliance processes within a single, configurable software platform. The system includes core quality and compliance workflows out of the box, eliminating the need for separate modules and reducing reliance on paper-based or disconnected systems.
Organizations can deploy the solutions they need today and add additional capabilities as requirements evolve. With pre-configured workflows, searchable data, and flexible integration options, AssurX supports efficient deployment, configuration, and long-term scalability across the enterprise.







