April 20, 2021
The number of U.S. Food and Drug Administration (FDA) inspections dropped dramatically in 2020, with the agency postponing most onsite visits to limit COVID-19 risks.
The number of FDA warning letters issued in 2020 kept pace with earlier years. This was due at least in part to the explosion of companies marketing questionable COVID-19 cures. Simultaneously, the number of 483 inspection observations fell nearly 42 percent in 2020 according to FDA data.
For example, the FDA sent over 2,700 Form 483 observation letters in 2020. This was compared with more than 4,700 in 2019, a 42% decrease. The trend is essentially the result of fewer inspections. However, compliance risks are in sharper focus for life sciences manufacturers with inspections resuming.
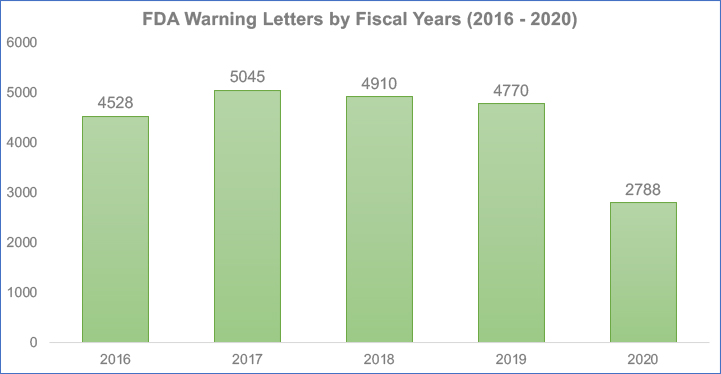
Source: FDA
What is the FDA likely to find after a year that has seen such seismic shifts in manufacturing? What issues are FDA inspectors likely to focus on? Most importantly, how can companies be ready?
Here we examine how 2020 has increased quality risks, how to achieve audit readiness, and how the enterprise quality management system (EQMS) can help.
Workplace Change and Risk for FDA Warning Letters and 483 Observations
Life sciences companies are significantly different today compared to the beginning of 2020. With many staff now working remotely, companies have fewer people on-site. There are also fewer personnel overall due to recession-driven job cuts.
The result for life sciences companies has been a loss of institutional knowledge and difficulty understanding data and making decisions. Ultimately, these problems can impact how companies detect and respond to issues, which may result in market withdrawals, FDA warning letters, and Form 483 observations.
For example, while essential workers have remained on site, many employees now work from home. If your quality group has gone from five people down to two, it only follows that those people may now have to deal with twice their average email volume. In this context, it may be likely that quality alerts or product issues could be missed. They could also have a delayed response.
Regaining FDA Audit-Readiness
A lot has happened in a year, and the key to avoiding FDA warning letters and Form 483 observations is audit readiness.
Specifically, you should be prioritizing your actions according to risk. Focus on identifying high-risk issues and putting them at the top of your to-do list. Areas to pay attention to include:
- Post-market surveillance: When it comes to managing complaints, it’s important not to focus exclusively on new products. It’s also essential to review complaints on older products to safeguard against any lurking problems.
- Managing essential workers: It’s critical to verify that your manufacturing process—which may have changed significantly—still meets quality standards. This may also mean evaluating gaps in your quality process due to teams connecting remotely vs. the way processes had been addressed in person.
- Supplier Quality: The pandemic has stretched the supply chain to its breaking point, forcing many companies to work with secondary and even tertiary suppliers. That requires new controls to protect against supplier quality issues—and documentation of the decisions your team had to make in the past year.
Documenting Your Decisions
As FDA inspections resume, it’s clear there will be much ground to cover after a yearlong hiatus. Having documentation for your decisions is key to avoiding findings. It also helps show you’ve done your due diligence when questions come up.
For example, let’s imagine you’re dealing with a shortage and receive two lots from different suppliers with differing levels of quality. Your primary supplier continues to provide what you want, but your backup supplier isn’t quite there yet.
Many companies in this position have had to make tough decisions, whether to deviate the product or reject the shipment and delay production. When FDA inspectors ask about it, you need to have findings documented, including objective evidence that backs up your rationale. If you run a planned deviation, for example, inspectors would want to know whether you assessed risks by:
- Monitoring post-market complaints related to this decision
- Identifying any changes in crucial quality trends arising from the material quality
- Looking for indications that the product affected the customer in any way
Use the EQMS to Respond to Questions
When the answers the FDA is looking for are buried in a folder, you’re much less likely to be able to quickly find them. This increases the risk you’ll receive a Form 483. Instead, you want to be able to say with confidence that you did not identify an impact. Present reports and charts as objective evidence to prove it.
The above scenario alone highlights multiple EQMS tools that would come into play, including:
- Document management
- Complaint handling
- Manufacturing quality records
- Supplier quality management
- Reporting
These aren’t the only areas of the EQMS critical to audit readiness, however. Life sciences companies also need to leverage tools such as:
- Training management: You should be able to pull up records showing that people have the right skills and certifications for their respective roles.
- Corrective and preventive action: Companies must provide details into root cause analysis findings and actions taken to resolve quality issues, as well as effectiveness checks.
- Audit management: For problems you’ve identified, FDA inspectors will want to see records that you continue to monitor the issue with internal audits.
- Change control: Changes to processes or equipment need to be documented and planned so as to minimize the impact on product quality and safety.
- Risk management: Being able to detect and identify high-risk quality items such as corrective actions, deviations, and internal audit findings allows you to proactively track issues likely to come up in an FDA inspection.
After a year of minimal onsite regulatory presence in life sciences manufacturing, it’s understandable that FDA warning letters and Form 483 observations will likely jump in 2021.
Hopefully, the FDA will recognize that companies today aren’t the same as they were the last time inspectors visited, providing extra leeway and guidance wherever possible. However, since we don’t know how the agency will approach resuming inspections, companies need to focus their energies on audit readiness and the digital tools that will get them there.


