August 15, 2017
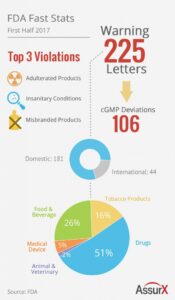
The FDA released 225 warning letters in the first half of the year. AssurX, a QMS software provider, provides a quick snapshot detailing the FDA's activity.
Top 3 FDA Warning Letter Violations
The top 3 FDA Warning Letter violations were (1) adulterated products, (2) unsanitary conditions, and (3) misbranded products. All three violations were applicable to both drugs and food.
Number of FDA Warning Letters
In the first half of 2017, a total of 225 Warning Letters were released. 181 domestic manufacturers received Warning Letters while 44 international manufacturers received Warning Letters. This correlates to approximately 80% of Warning Letters issued to domestic manufacturers while 20% of the FDA Warning Letters issued by inspectors were to international manufacturers.
Also of note were 106 cGMP deviations recorded.
Manufacturer Type Breakdown
The breakdown of manufacturer type receiving FDA Warning Letters:
- 51% Drug Manufacturers
- 26% Food & Beverage Manufacturers
- 16% Tobacco Product Manufacturers
- 5% Medical Device Manufacturers
- 2% Animal & Veterinary Product Manufacturers
Key Insights
- The FDA continues to follow complaints across all manufacturing entities and is clearly taking those complaints seriously.
- Drugs including API and supplement manufacturers received more than half of all warning letters.
- Supplement manufacturers have received a slew of letters for unvalidated scientific claims about the healing properties of their "natural" product ingredients.
- The vaping/e-cigarette industry is now feeling the pain of being subject to new rules added on August 8, 2016, to the Tobacco Control Act.
- Insanitary seafood preparation and packing still remain a major HACCP violation.
Conclusion
In the calendar year 2016, the overall number of quality management system surveillance inspections by the FDA was slightly higher. Certain citation patterns are certainly emerging in 2017. Will the 80/20 warning letter breakdown between domestic and international manufacturers continue for the rest of 2017?